Novatend’s cutting-edge tissue engineering platform provides a full suite of bone grafting and biomaterial solutions designed to meet your surgical needs.
Learn About Us...
NEXT-GENENRATION
TISSUE-ENGINEERING
Novatend leverages its expertise in tissue engineering and medical device manufacturing to deliver the most affordable bone grafting solutions direct to customer. Proud partner of Altis Biologics (Pty) Ltd.
LEARN MORE

MOST POPULAR DEVICES
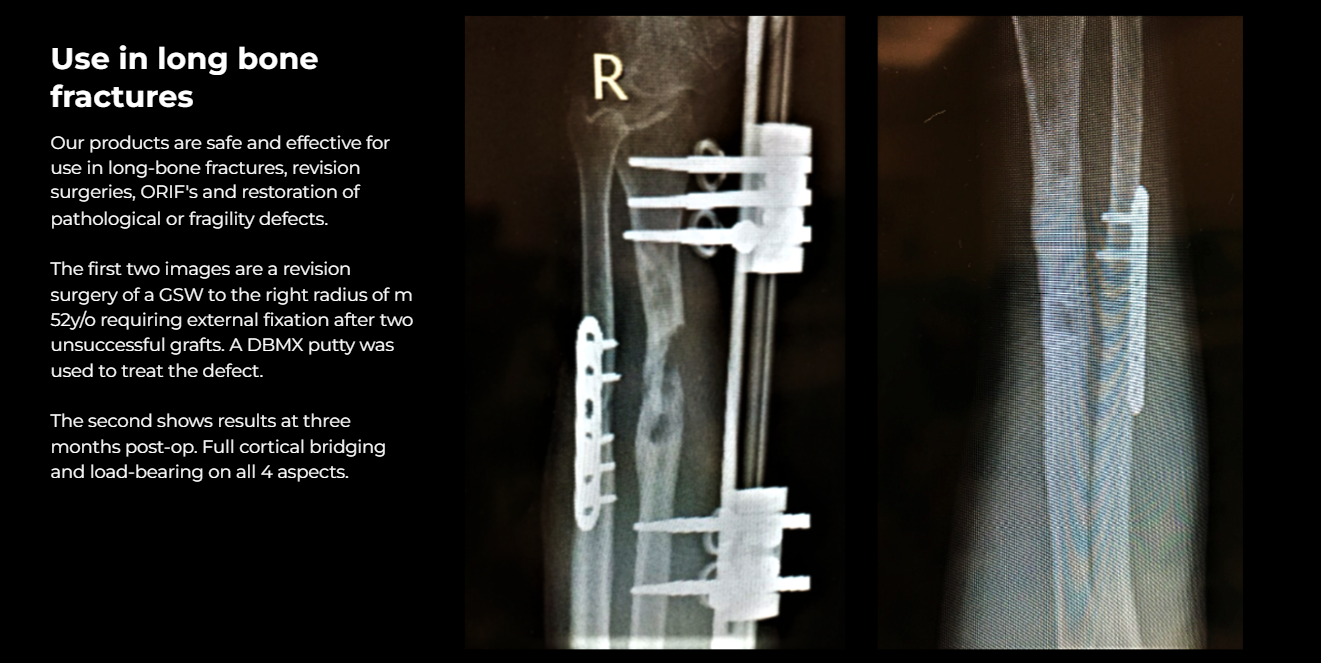
All devices are high-quality locally manufactured grafts that yield predictable and reliable clinical outcomes.

NovaDBMX
Ready-to-inject osteoinductive putty containing high levels of BMP-complex for spinal fusions, ORIFS and non-unions

NovaGROW
DBM Granules with standardized BMP levels for sinus lifts, ridge augmentations and socket preservations

NovaMEM
Atelocollagen barrier membrane for graft sequestration/tissue approximation

NovaGRAFT
Biocompatible spongiosa for reconstructive procedures

NovatendX
Bioresorbable rotator cuff patch graft for the augmentation of traditional repairs

NovaSGX
Fully synthetic ready-to-inject graft for maxillofacial and periodontal procedures
NEXT-GEN INJECTABLE
BONE GRAFTING
We provide the latest in bone grafting materials straight to our customers. This enables shorter time spent in theater, reduced risk due to disease-transfer and infection and for a fraction of the price of a second procedure
Get Started

We have built a great platform in which we can provide cutting edge regenerative medicine based solutions directly to distributors, hospital groups and surgeons. Our devices sell at a fraction of the cost of imported competitors and are fully reimbursed by major medical aid groups, giving patients a much needed break from inflated out-of-pocket expenses.
- MC Labuschagne, Executive Director
2241
Succesful Operations
16.4%
Month on Month Growth
Q1-Q4 2025
0
Adverse Events Reported